Seznamy Atom Labeled Nucleus Zdarma
Seznamy Atom Labeled Nucleus Zdarma. Because of variations in binding energy, some nuclei are unstable and … Nucleus occupies an extremely small volume inside the atom. Atoms have different properties based on the arrangement and number of their basic particles.
Prezentováno Rutherford Model Of The Atom Definition Diagram Video Lesson Transcript Study Com
The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The volume of a nucleus was considered to be negligible compared to the volume of an atom. An atom has a central nucleus. The binding energy of a nucleus is the energy holding a nucleus together. 10 november 2020 * if you want to update the article please login/register.The following article provides you with diagrams that will help you understand the structure of atom better.
Nucleus occupies an extremely small volume inside the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells. Rutherford compared his atom …

It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together. The nucleus is tiny compared to the atom as a whole: It is a neutral particle. This is surrounded by electrons arranged in shells. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The atom had a lot of empty space. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Nucleus occupies an extremely small volume inside the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

An atom has a central nucleus... The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Rutherford compared his atom … According to his atom diagram, the atom has a small, positively charged nucleus in center. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows: Furthermore, protons and neutrons have a mass that is nearly 1 amu.

Because of variations in binding energy, some nuclei are unstable and … The nucleus is tiny compared to the atom as a whole: Nucleus occupies an extremely small volume inside the atom. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells.. Atoms have different properties based on the arrangement and number of their basic particles.

The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows: Nucleus occupies an extremely small volume inside the atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. 10 november 2020 * if you want to update the article please login/register. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Labeled atom summarized by plexpage. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Because of variations in binding energy, some nuclei are unstable and … The volume of a nucleus was considered to be negligible compared to the volume of an atom... The nucleus is tiny compared to the atom as a whole:
Atom is basic unit of matter. 10 november 2020 * if you want to update the article please login/register. An atom has a central nucleus. The atom had a lot of empty space. The nucleus is tiny compared to the atom as a whole: The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells. Labeled atom summarized by plexpage. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells.

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. Because of variations in binding energy, some nuclei are unstable and … The binding energy of a nucleus is the energy holding a nucleus together. An atom has a central nucleus. This nucleus carries the entire mass of the atom. Labeled atom summarized by plexpage. Nucleus occupies an extremely small volume inside the atom. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The following article provides you with diagrams that will help you understand the structure of atom better. But dalton or amu (atomic mass unit) is appropriate. The binding energy of a nucleus is the energy holding a nucleus together.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Atom is basic unit of matter. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. This nucleus carries the entire mass of the atom. Because of variations in binding energy, some nuclei are unstable and … Atoms have different properties based on the arrangement and number of their basic particles. The following article provides you with diagrams that will help you understand the structure of atom better. But dalton or amu (atomic mass unit) is appropriate. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells.

Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Labeled atom summarized by plexpage. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

According to his atom diagram, the atom has a small, positively charged nucleus in center.. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Atoms have different properties based on the arrangement and number of their basic particles. Rutherford compared his atom … 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. This nucleus carries the entire mass of the atom. It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together. But dalton or amu (atomic mass unit) is appropriate. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The volume of a nucleus was considered to be negligible compared to the volume of an atom.

10 november 2020 * if you want to update the article please login/register. Atom is basic unit of matter. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The following article provides you with diagrams that will help you understand the structure of atom better.

Nucleus occupies an extremely small volume inside the atom... . The nucleus is tiny compared to the atom as a whole:

The atom had a lot of empty space. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. Nucleus occupies an extremely small volume inside the atom. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). The atom had a lot of empty space. The binding energy of a nucleus is the energy holding a nucleus together. Labeled atom summarized by plexpage. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons.

Atoms have different properties based on the arrangement and number of their basic particles. This is surrounded by electrons arranged in shells. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Furthermore, protons and neutrons have a mass that is nearly 1 amu. The following article provides you with diagrams that will help you understand the structure of atom better. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). According to his atom diagram, the atom has a small, positively charged nucleus in center. 10 november 2020 * if you want to update the article please login/register. Nucleus occupies an extremely small volume inside the atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.. It is a neutral particle.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number... The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). It is a neutral particle. 10 november 2020 * if you want to update the article please login/register. This nucleus carries the entire mass of the atom. The binding energy of a nucleus is the energy holding a nucleus together. This is surrounded by electrons arranged in shells... 10 november 2020 * if you want to update the article please login/register.

The following article provides you with diagrams that will help you understand the structure of atom better. This nucleus carries the entire mass of the atom. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). It is a neutral particle. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Furthermore, protons and neutrons have a mass that is nearly 1 amu. This is surrounded by electrons arranged in shells.. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

Rutherford compared his atom …. But dalton or amu (atomic mass unit) is appropriate. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The atom had a lot of empty space. Nucleus occupies an extremely small volume inside the atom. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). An atom has a central nucleus... The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.

Because of variations in binding energy, some nuclei are unstable and …. The nucleus is tiny compared to the atom as a whole: Because of variations in binding energy, some nuclei are unstable and … It is a neutral particle. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The following article provides you with diagrams that will help you understand the structure of atom better. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

This nucleus carries the entire mass of the atom. Nucleus occupies an extremely small volume inside the atom. This is surrounded by electrons arranged in shells. This nucleus carries the entire mass of the atom. It is a neutral particle. 10 november 2020 * if you want to update the article please login/register.

10 november 2020 * if you want to update the article please login/register... 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The binding energy of a nucleus is the energy holding a nucleus together. It is a neutral particle. The atom had a lot of empty space.. 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows:

According to his atom diagram, the atom has a small, positively charged nucleus in center. 10 november 2020 * if you want to update the article please login/register. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). An atom has a central nucleus. Rutherford compared his atom … 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Atom is basic unit of matter. It is a neutral particle.. But dalton or amu (atomic mass unit) is appropriate.

The nucleus is tiny compared to the atom as a whole:.. The following article provides you with diagrams that will help you understand the structure of atom better. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Gram is not an appropriate unit for the calculation of such tiny subatomic particles. An atom has a central nucleus. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nucleus is tiny compared to the atom as a whole: Nucleus occupies an extremely small volume inside the atom. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).. According to his atom diagram, the atom has a small, positively charged nucleus in center.

The following article provides you with diagrams that will help you understand the structure of atom better.. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). According to his atom diagram, the atom has a small, positively charged nucleus in center. This is surrounded by electrons arranged in shells.. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells.
The binding energy of a nucleus is the energy holding a nucleus together. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Atom is basic unit of matter. Furthermore, protons and neutrons have a mass that is nearly 1 amu.. Atoms have different properties based on the arrangement and number of their basic particles.

This is surrounded by electrons arranged in shells. The atom had a lot of empty space. The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom.. This nucleus carries the entire mass of the atom.

Furthermore, protons and neutrons have a mass that is nearly 1 amu. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. 10 november 2020 * if you want to update the article please login/register. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Because of variations in binding energy, some nuclei are unstable and … The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). An atom has a central nucleus. 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows: This nucleus carries the entire mass of the atom. It is a neutral particle. The nucleus is tiny compared to the atom as a whole:.. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

This is surrounded by electrons arranged in shells. Because of variations in binding energy, some nuclei are unstable and … Rutherford compared his atom … It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Atoms have different properties based on the arrangement and number of their basic particles. This is surrounded by electrons arranged in shells. 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows:. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons.
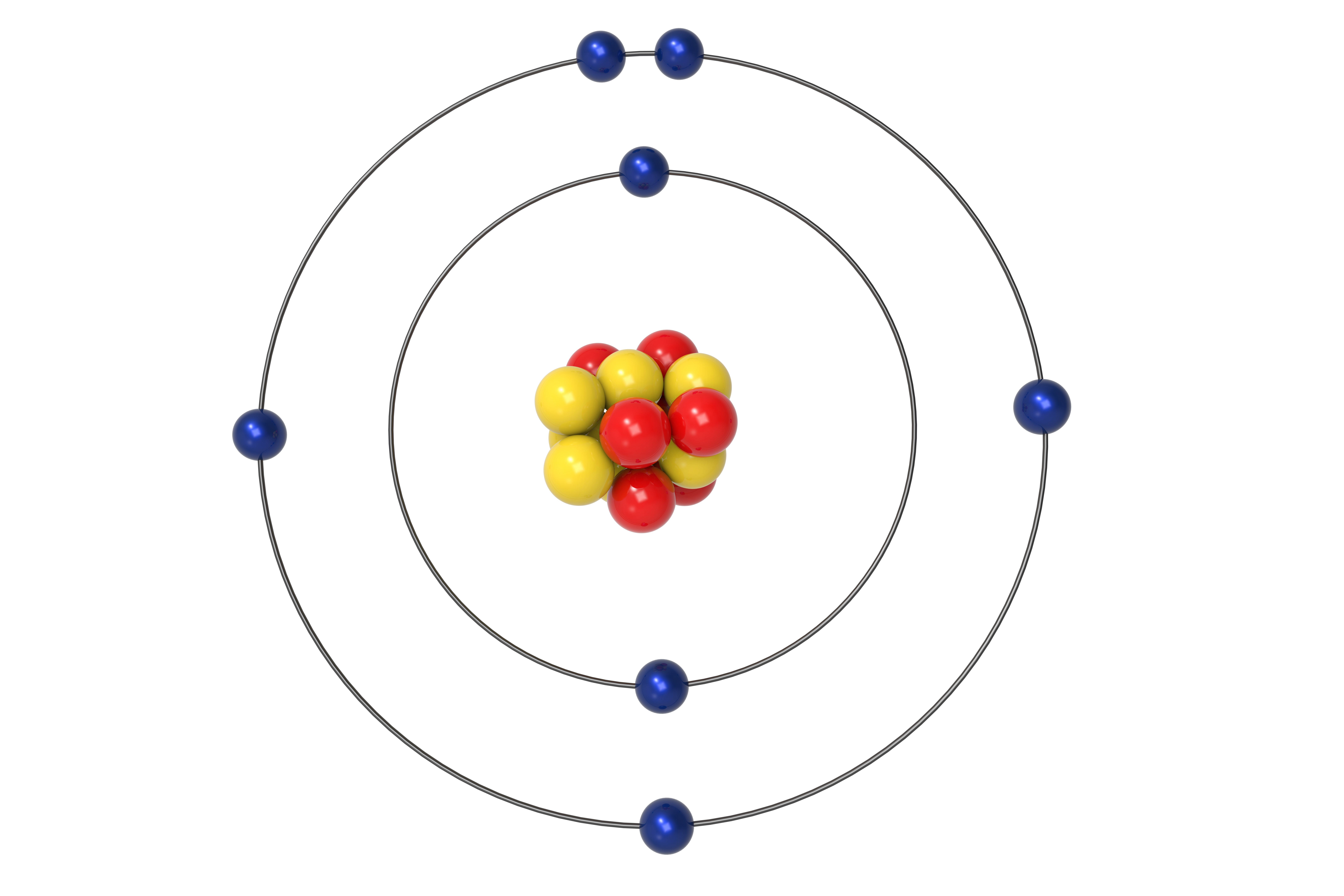
The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Atom is basic unit of matter. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together. The nucleus is tiny compared to the atom as a whole: But dalton or amu (atomic mass unit) is appropriate. According to his atom diagram, the atom has a small, positively charged nucleus in center. This nucleus carries the entire mass of the atom. This is surrounded by electrons arranged in shells. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.

But dalton or amu (atomic mass unit) is appropriate.. It is a neutral particle.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The following article provides you with diagrams that will help you understand the structure of atom better. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The binding energy of a nucleus is the energy holding a nucleus together... The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.
Furthermore, protons and neutrons have a mass that is nearly 1 amu... . 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.

10 november 2020 * if you want to update the article please login/register. The volume of a nucleus was considered to be negligible compared to the volume of an atom. An atom has a central nucleus. This nucleus carries the entire mass of the atom. Atom is basic unit of matter. Atoms have different properties based on the arrangement and number of their basic particles. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Labeled atom summarized by plexpage. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells. It is a neutral particle. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

This is surrounded by electrons arranged in shells.. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Nucleus occupies an extremely small volume inside the atom. Atom is basic unit of matter. Atoms have different properties based on the arrangement and number of their basic particles. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.

10 november 2020 * if you want to update the article please login/register. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). It is a neutral particle. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The nucleus is tiny compared to the atom as a whole: Nucleus occupies an extremely small volume inside the atom. But dalton or amu (atomic mass unit) is appropriate.

This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. But dalton or amu (atomic mass unit) is appropriate. This nucleus carries the entire mass of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. This is surrounded by electrons arranged in shells. Atom is basic unit of matter. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Furthermore, protons and neutrons have a mass that is nearly 1 amu. Labeled atom summarized by plexpage.. It is a neutral particle.

According to his atom diagram, the atom has a small, positively charged nucleus in center. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Rutherford compared his atom … The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). This is surrounded by electrons arranged in shells. This nucleus carries the entire mass of the atom. The following article provides you with diagrams that will help you understand the structure of atom better.

According to his atom diagram, the atom has a small, positively charged nucleus in center... The following article provides you with diagrams that will help you understand the structure of atom better. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The nucleus of an atom usually contains the proton and the neutron while the electrons are found outside the nucleus within the electron shells. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number... The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).
24.09.2019 · the parts of the atom that are labeled in the diagram are as follows:. The atom had a lot of empty space. Nucleus occupies an extremely small volume inside the atom. But dalton or amu (atomic mass unit) is appropriate. An atom has a central nucleus. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Because of variations in binding energy, some nuclei are unstable and … The volume of a nucleus was considered to be negligible compared to the volume of an atom.. Nucleus occupies an extremely small volume inside the atom.

10 november 2020 * if you want to update the article please login/register. 10 november 2020 * if you want to update the article please login/register. Atoms have different properties based on the arrangement and number of their basic particles. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Rutherford compared his atom … The following article provides you with diagrams that will help you understand the structure of atom better. This is surrounded by electrons arranged in shells.. This nucleus carries the entire mass of the atom.

Rutherford compared his atom … Because of variations in binding energy, some nuclei are unstable and … The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. 10 november 2020 * if you want to update the article please login/register. It is a neutral particle. Atoms have different properties based on the arrangement and number of their basic particles. The atom had a lot of empty space. This is surrounded by electrons arranged in shells... 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.

Labeled atom summarized by plexpage.. 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows: This is surrounded by electrons arranged in shells. Atom is basic unit of matter.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. 24.09.2019 · the parts of the atom that are labeled in the diagram are as follows: It is a neutral particle. But dalton or amu (atomic mass unit) is appropriate. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

Atom is basic unit of matter... This is surrounded by electrons arranged in shells. The nucleus is tiny compared to the atom as a whole: Rutherford compared his atom ….. It is a neutral particle.

But dalton or amu (atomic mass unit) is appropriate.. 10 november 2020 * if you want to update the article please login/register. This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The nucleus is tiny compared to the atom as a whole: The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. But dalton or amu (atomic mass unit) is appropriate. An atom has a central nucleus. Atom is basic unit of matter. According to his atom diagram, the atom has a small, positively charged nucleus in center.. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons.

Atoms have different properties based on the arrangement and number of their basic particles.. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.. But dalton or amu (atomic mass unit) is appropriate. Nucleus occupies an extremely small volume inside the atom. Rutherford compared his atom … 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. According to his atom diagram, the atom has a small, positively charged nucleus in center. An atom has a central nucleus. The atom is the smallest building block of all matter made up of neutrons, protons, and electrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). But dalton or amu (atomic mass unit) is appropriate.
