Kolekce 187+ Classical Solar System Atomic Model Čerstvý
Kolekce 187+ Classical Solar System Atomic Model Čerstvý. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in …
Prezentováno Atomic Structure Notes Ppt Video Online Download
The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. 28/11/2015 · it is a classical physics' problem! The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).28/11/2015 · it is a classical physics' problem!
It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … 4.4 the bohr model of the hydrogen atom; In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …
It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly ….. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was a danish scientist who is best known for his contributions to the atomic model. Free online model of solar system and night sky. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 28/11/2015 · it is a classical physics' problem!

It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. 4.4 the bohr model of the hydrogen atom; 28/11/2015 · it is a classical physics' problem! Who invented the solar system model of the atom? The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. He was the first to realize that electrons travel in … 4.3 the instability of the classic "solar system" model of atoms. Free online model of solar system and night sky.
4.4 the bohr model of the hydrogen atom; 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus ….. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …

08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom.. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Free online model of solar system and night sky. Bohr, one of the pioneers of … 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,.
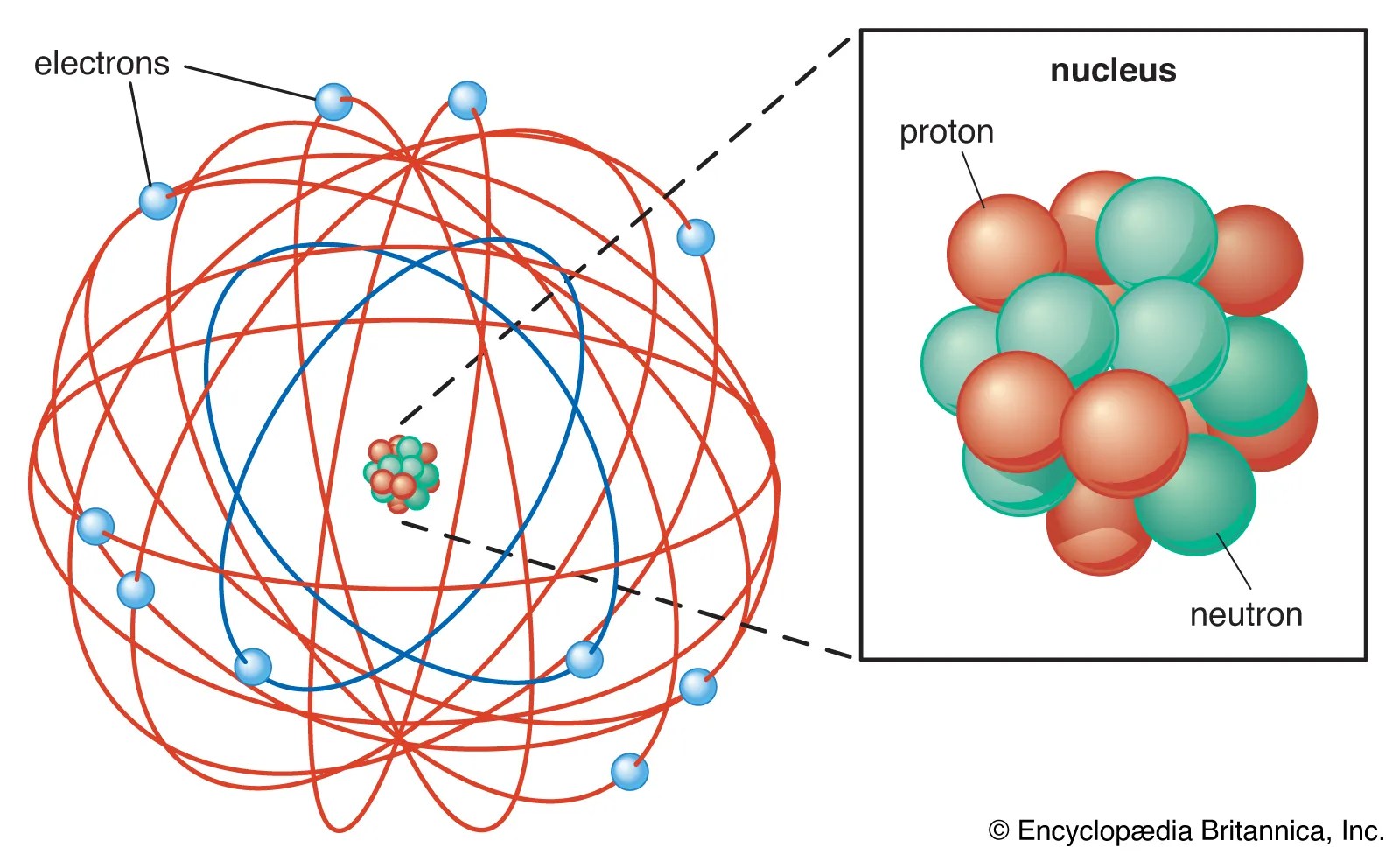
The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … Bohr, one of the pioneers of … But… in 1912 niels bohr pointed out that the orbi.

08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. Who invented the solar system model of the atom?.. To run the online …

Contact@solarsystemscope.com facebook newsletter embed account. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. Contact@solarsystemscope.com facebook newsletter embed account. 4.3 the instability of the classic "solar system" model of atoms. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …. Who invented the solar system model of the atom?

The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. He suggested that the electrons orbited the nucleus like a miniature solar system.

But… in 1912 niels bohr pointed out that the orbi... Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 4.3 the instability of the classic "solar system" model of atoms. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 4.4 the bohr model of the hydrogen atom; He was the first to realize that electrons travel in … To run the online … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …

The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. Neils bohr came up the solar system model of the atom in 1913. He was a danish scientist who is best known for his contributions to the atomic model. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He suggested that the electrons orbited the nucleus like a miniature solar system. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 28/11/2015 · it is a classical physics' problem! Bohr, one of the pioneers of …. Contact@solarsystemscope.com facebook newsletter embed account.

Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,... The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Who invented the solar system model of the atom? To run the online … The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... 4.3 the instability of the classic "solar system" model of atoms.

4.3 the instability of the classic "solar system" model of atoms. Contact@solarsystemscope.com facebook newsletter embed account. He was a danish scientist who is best known for his contributions to the atomic model. Who invented the solar system model of the atom? It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.
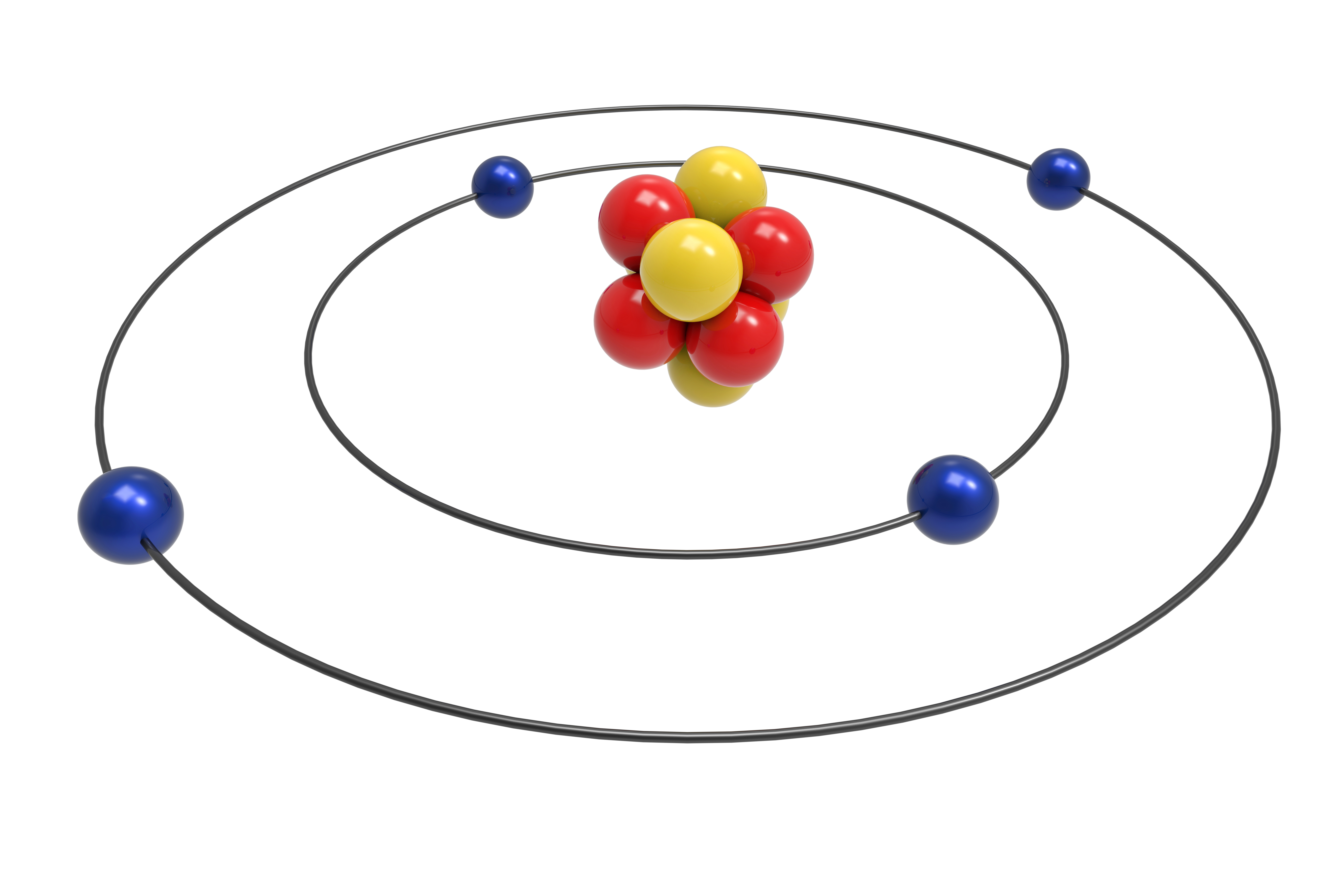
28/11/2015 · it is a classical physics' problem!.. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. 4.4 the bohr model of the hydrogen atom; 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. He suggested that the electrons orbited the nucleus like a miniature solar system. Neils bohr came up the solar system model of the atom in 1913.. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.

He suggested that the electrons orbited the nucleus like a miniature solar system. Neils bohr came up the solar system model of the atom in 1913. Bohr, one of the pioneers of … 28/11/2015 · it is a classical physics' problem! 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Contact@solarsystemscope.com facebook newsletter embed account. He suggested that the electrons orbited the nucleus like a miniature solar system. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom.. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.

12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. Free online model of solar system and night sky. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. 28/11/2015 · it is a classical physics' problem! In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.

08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom.. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Bohr, one of the pioneers of … It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …

Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,.. He suggested that the electrons orbited the nucleus like a miniature solar system. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.. He was the first to realize that electrons travel in …
4.4 the bohr model of the hydrogen atom; Bohr, one of the pioneers of … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. 28/11/2015 · it is a classical physics' problem! 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. But… in 1912 niels bohr pointed out that the orbi. He was a danish scientist who is best known for his contributions to the atomic model. Contact@solarsystemscope.com facebook newsletter embed account. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly ….. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.

It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … 4.3 the instability of the classic "solar system" model of atoms. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. Who invented the solar system model of the atom? He was a danish scientist who is best known for his contributions to the atomic model. Bohr, one of the pioneers of … Neils bohr came up the solar system model of the atom in 1913. To run the online ….. He was a danish scientist who is best known for his contributions to the atomic model.
The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.. 4.3 the instability of the classic "solar system" model of atoms... Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,.

4.3 the instability of the classic "solar system" model of atoms.. But… in 1912 niels bohr pointed out that the orbi. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant... Bohr, one of the pioneers of …

To run the online … The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. But… in 1912 niels bohr pointed out that the orbi. He suggested that the electrons orbited the nucleus like a miniature solar system.

He was the first to realize that electrons travel in ….. He was the first to realize that electrons travel in … Neils bohr came up the solar system model of the atom in 1913. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. To run the online … 4.3 the instability of the classic "solar system" model of atoms. But… in 1912 niels bohr pointed out that the orbi.. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.

3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. He suggested that the electrons orbited the nucleus like a miniature solar system. Bohr, one of the pioneers of … It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun... In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass.

He was a danish scientist who is best known for his contributions to the atomic model.. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. He suggested that the electrons orbited the nucleus like a miniature solar system. 4.3 the instability of the classic "solar system" model of atoms.. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass.
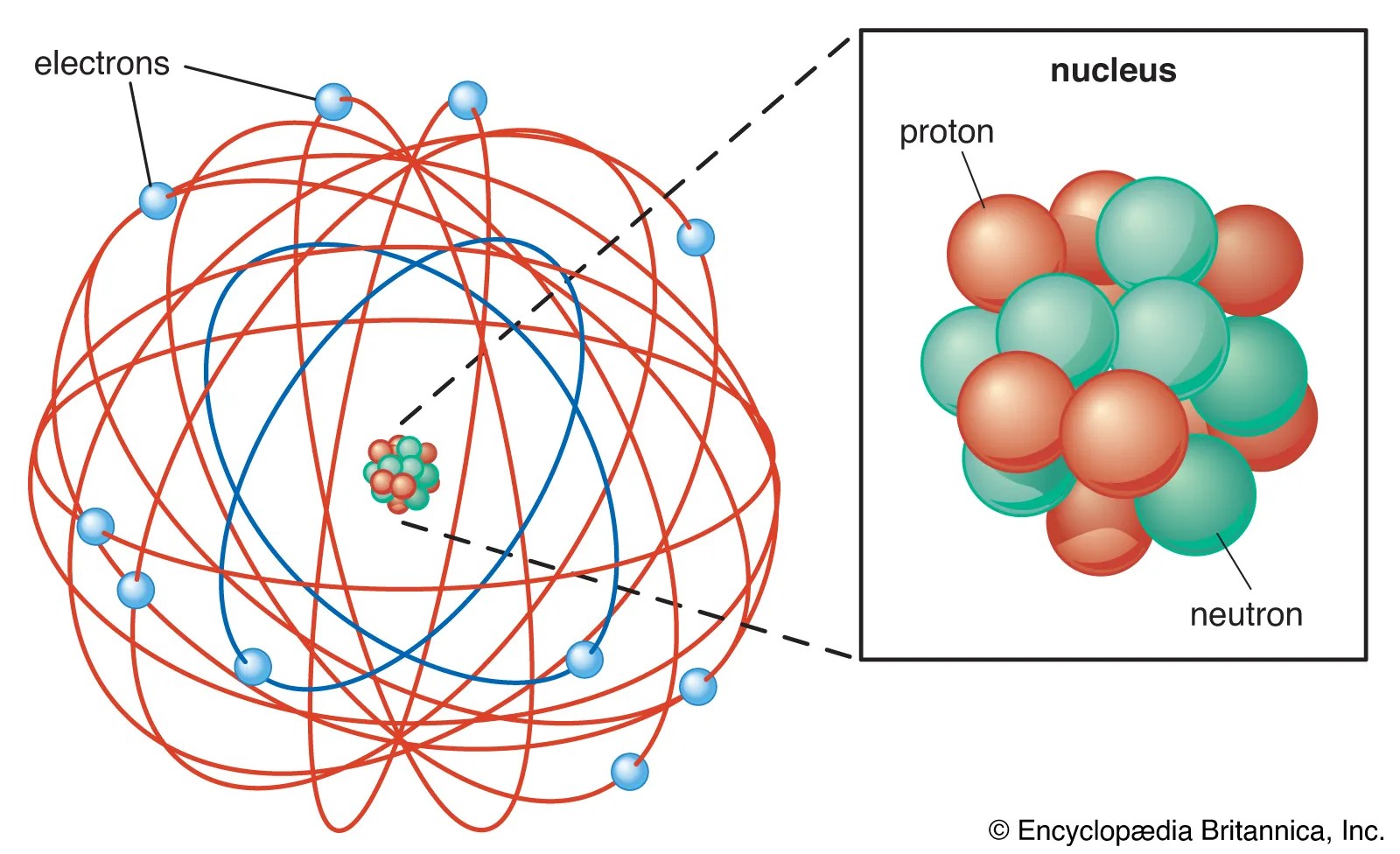
But… in 1912 niels bohr pointed out that the orbi. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 4.3 the instability of the classic "solar system" model of atoms. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Bohr, one of the pioneers of … He suggested that the electrons orbited the nucleus like a miniature solar system. But… in 1912 niels bohr pointed out that the orbi. Neils bohr came up the solar system model of the atom in 1913. He was the first to realize that electrons travel in …

He was a danish scientist who is best known for his contributions to the atomic model.. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Neils bohr came up the solar system model of the atom in 1913. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. 28/11/2015 · it is a classical physics' problem! Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom.

Who invented the solar system model of the atom? The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. 4.4 the bohr model of the hydrogen atom; 4.3 the instability of the classic "solar system" model of atoms. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun.

3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. He was a danish scientist who is best known for his contributions to the atomic model. Bohr, one of the pioneers of … Who invented the solar system model of the atom? Neils bohr came up the solar system model of the atom in 1913. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). But… in 1912 niels bohr pointed out that the orbi.. He was the first to realize that electrons travel in …
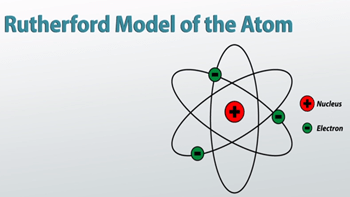
28/11/2015 · it is a classical physics' problem! The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. To run the online … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). 4.3 the instability of the classic "solar system" model of atoms. Bohr, one of the pioneers of … The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. Neils bohr came up the solar system model of the atom in 1913.. But… in 1912 niels bohr pointed out that the orbi.

Contact@solarsystemscope.com facebook newsletter embed account. Neils bohr came up the solar system model of the atom in 1913. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. Contact@solarsystemscope.com facebook newsletter embed account. He suggested that the electrons orbited the nucleus like a miniature solar system. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Free online model of solar system and night sky. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of... Who invented the solar system model of the atom?

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. Who invented the solar system model of the atom? 28/11/2015 · it is a classical physics' problem! To run the online … The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. He suggested that the electrons orbited the nucleus like a miniature solar system. 4.3 the instability of the classic "solar system" model of atoms.

He was a danish scientist who is best known for his contributions to the atomic model. 28/11/2015 · it is a classical physics' problem! The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … Free online model of solar system and night sky.. 4.4 the bohr model of the hydrogen atom;

4.4 the bohr model of the hydrogen atom;. Contact@solarsystemscope.com facebook newsletter embed account. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. But… in 1912 niels bohr pointed out that the orbi. Free online model of solar system and night sky. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). To run the online … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … 4.4 the bohr model of the hydrogen atom; In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. Contact@solarsystemscope.com facebook newsletter embed account. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.
The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. Free online model of solar system and night sky.. Neils bohr came up the solar system model of the atom in 1913.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. He was the first to realize that electrons travel in … 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.

But… in 1912 niels bohr pointed out that the orbi. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. But… in 1912 niels bohr pointed out that the orbi.. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).

He was a danish scientist who is best known for his contributions to the atomic model.. 28/11/2015 · it is a classical physics' problem!. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.

Neils bohr came up the solar system model of the atom in 1913. 4.3 the instability of the classic "solar system" model of atoms. Contact@solarsystemscope.com facebook newsletter embed account. Who invented the solar system model of the atom? In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. Neils bohr came up the solar system model of the atom in 1913.

Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,... He was the first to realize that electrons travel in … Contact@solarsystemscope.com facebook newsletter embed account. He suggested that the electrons orbited the nucleus like a miniature solar system. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Who invented the solar system model of the atom? The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Neils bohr came up the solar system model of the atom in 1913. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.
Free online model of solar system and night sky. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Free online model of solar system and night sky. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass... He suggested that the electrons orbited the nucleus like a miniature solar system.

He was the first to realize that electrons travel in … Contact@solarsystemscope.com facebook newsletter embed account. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). 4.3 the instability of the classic "solar system" model of atoms. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. Bohr, one of the pioneers of …. Free online model of solar system and night sky.

Bohr, one of the pioneers of ….. 4.3 the instability of the classic "solar system" model of atoms. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).

3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant... It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … 28/11/2015 · it is a classical physics' problem! Who invented the solar system model of the atom? Free online model of solar system and night sky.

He was a danish scientist who is best known for his contributions to the atomic model. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Contact@solarsystemscope.com facebook newsletter embed account. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. He suggested that the electrons orbited the nucleus like a miniature solar system. Who invented the solar system model of the atom? Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … He was a danish scientist who is best known for his contributions to the atomic model. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... Free online model of solar system and night sky.

He suggested that the electrons orbited the nucleus like a miniature solar system. He was the first to realize that electrons travel in … Contact@solarsystemscope.com facebook newsletter embed account. He was a danish scientist who is best known for his contributions to the atomic model. Who invented the solar system model of the atom? The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Bohr, one of the pioneers of … 4.3 the instability of the classic "solar system" model of atoms. Free online model of solar system and night sky. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom.

4.4 the bohr model of the hydrogen atom; Who invented the solar system model of the atom? The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. He was the first to realize that electrons travel in … Free online model of solar system and night sky. Contact@solarsystemscope.com facebook newsletter embed account.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). 28/11/2015 · it is a classical physics' problem! Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. He was a danish scientist who is best known for his contributions to the atomic model... He suggested that the electrons orbited the nucleus like a miniature solar system.
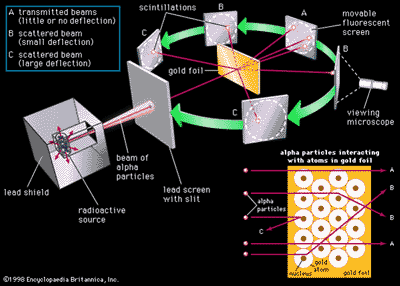
The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. Contact@solarsystemscope.com facebook newsletter embed account. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. Free online model of solar system and night sky. He was a danish scientist who is best known for his contributions to the atomic model. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly ….. But… in 1912 niels bohr pointed out that the orbi.

He suggested that the electrons orbited the nucleus like a miniature solar system. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. But… in 1912 niels bohr pointed out that the orbi. Contact@solarsystemscope.com facebook newsletter embed account. 4.3 the instability of the classic "solar system" model of atoms. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … Neils bohr came up the solar system model of the atom in 1913. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … 28/11/2015 · it is a classical physics' problem! The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. To run the online …

The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. Free online model of solar system and night sky. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … He was the first to realize that electrons travel in … The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. But… in 1912 niels bohr pointed out that the orbi.. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

Bohr, one of the pioneers of … .. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets).

In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass... Contact@solarsystemscope.com facebook newsletter embed account. To run the online … But… in 1912 niels bohr pointed out that the orbi. Bohr, one of the pioneers of …. Contact@solarsystemscope.com facebook newsletter embed account.

He was the first to realize that electrons travel in … Free online model of solar system and night sky. Contact@solarsystemscope.com facebook newsletter embed account. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Bohr, one of the pioneers of … 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … But… in 1912 niels bohr pointed out that the orbi.

It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly ….. He was the first to realize that electrons travel in … 28/11/2015 · it is a classical physics' problem! 4.3 the instability of the classic "solar system" model of atoms. But… in 1912 niels bohr pointed out that the orbi.

The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Contact@solarsystemscope.com facebook newsletter embed account. Who invented the solar system model of the atom? 28/11/2015 · it is a classical physics' problem! He was the first to realize that electrons travel in ….. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

Who invented the solar system model of the atom?.. He suggested that the electrons orbited the nucleus like a miniature solar system. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. 4.3 the instability of the classic "solar system" model of atoms. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.

The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.. 4.4 the bohr model of the hydrogen atom; To run the online ….. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets)... 4.4 the bohr model of the hydrogen atom; 28/11/2015 · it is a classical physics' problem! 4.3 the instability of the classic "solar system" model of atoms. Bohr, one of the pioneers of … The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant... Bohr, one of the pioneers of …

To run the online … .. Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,.

In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom... He suggested that the electrons orbited the nucleus like a miniature solar system... 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.

The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). Who invented the solar system model of the atom? 28/11/2015 · it is a classical physics' problem! Free online model of solar system and night sky. Bohr, one of the pioneers of … Contact@solarsystemscope.com facebook newsletter embed account. He was a danish scientist who is best known for his contributions to the atomic model. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.

Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. But… in 1912 niels bohr pointed out that the orbi.. Free online model of solar system and night sky.

The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of.. He was the first to realize that electrons travel in …. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.
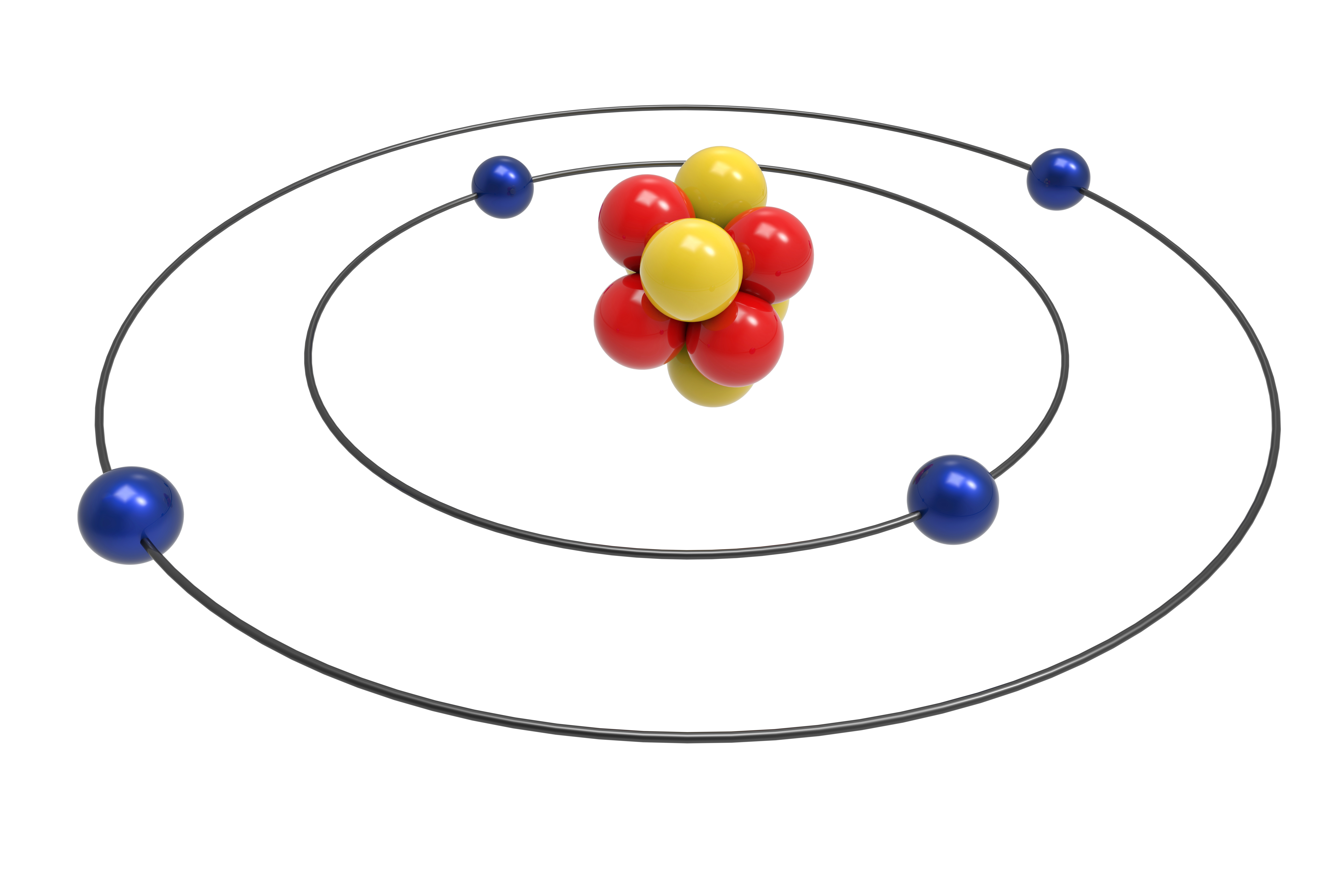
Who invented the solar system model of the atom? .. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus …

The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. 4.3 the instability of the classic "solar system" model of atoms. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. To run the online ….. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.

3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. Free online model of solar system and night sky. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. The problem with this is that the electrons are charged particles and moving around in a circle they have centripetal acceleration (even if they move with constant velocity in modulus … Neils bohr came up the solar system model of the atom in 1913. 4.3 the instability of the classic "solar system" model of atoms. 28/11/2015 · it is a classical physics' problem!. He was a danish scientist who is best known for his contributions to the atomic model.

In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass... Who invented the solar system model of the atom? In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. Contact@solarsystemscope.com facebook newsletter embed account. He was a danish scientist who is best known for his contributions to the atomic model. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. Neils bohr came up the solar system model of the atom in 1913. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …
The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits... 4.4 the bohr model of the hydrogen atom; Who invented the solar system model of the atom? Bohr, one of the pioneers of … He was the first to realize that electrons travel in … Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. 4.3 the instability of the classic "solar system" model of atoms.. 4.3 the instability of the classic "solar system" model of atoms.

Free online model of solar system and night sky. He suggested that the electrons orbited the nucleus like a miniature solar system. Contact@solarsystemscope.com facebook newsletter embed account. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. Neils bohr came up the solar system model of the atom in 1913. Free online model of solar system and night sky.
The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.. Bohr, one of the pioneers of …

In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom... 28/11/2015 · it is a classical physics' problem! 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. 4.3 the instability of the classic "solar system" model of atoms. 4.4 the bohr model of the hydrogen atom; In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. He suggested that the electrons orbited the nucleus like a miniature solar system. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly …

In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom.. He was the first to realize that electrons travel in … It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … To run the online … But… in 1912 niels bohr pointed out that the orbi. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom... 4.3 the instability of the classic "solar system" model of atoms.
Neils bohr came up the solar system model of the atom in 1913.. Who invented the solar system model of the atom?

To run the online … Contact@solarsystemscope.com facebook newsletter embed account. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. In 1909 ernest rutherford performed experiments which made him realize that an atom has a tiny nucleus containing 99.99% of the atom's mass. Bohr, one of the pioneers of … 4.3 the instability of the classic "solar system" model of atoms. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant.. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits.
12/07/2013 · the most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. Free online model of solar system and night sky. 4.4 the bohr model of the hydrogen atom; Solar absorption lines are called fraunhofer lines after joseph von fraunhofer,. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. The solar system model describes an atom as a central massive positive entity (the nucleus/sun) and, orbiting around it, the negative entities (the electrons/planets). 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. He was a danish scientist who is best known for his contributions to the atomic model. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of.
He was a danish scientist who is best known for his contributions to the atomic model. 3.3 line spectra of elements, balmer's numerology and rydberg's equation and constant. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly … 4.4 the bohr model of the hydrogen atom; The remarkable success of this model prompted many physicists to seek an explanation for why such a model should work at all, and to seek an understanding of. The rutherford atomic model placed the protons and neutrons inside a small structure called the 'nucleus' with the electrons revolving around the nucleus in circular orbits. In july of 1913, danish physicist niels bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the bohr atom. 08/07/2020 · with his classic, alpha ray scattering experiment rutherford was able to suggest a possible model for the atom. Contact@solarsystemscope.com facebook newsletter embed account. 28/11/2015 · it is a classical physics' problem!.. 4.4 the bohr model of the hydrogen atom;